Using Electrochemistry to Understand and Modulate Biological Activity
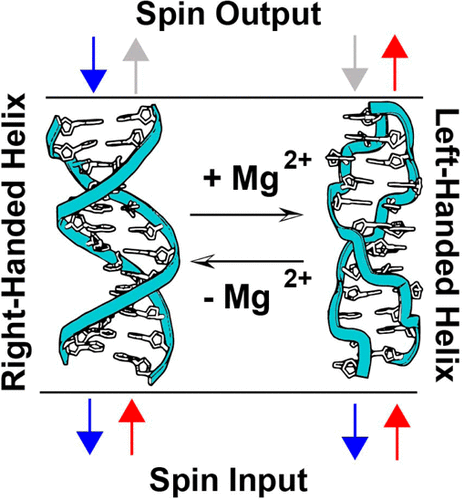
I built an electrochemical setup which allows for a spin polarized current to be injected from a ferromagnetic nickel layer into DNA. I found that the direction the nickel was magnetized controlled the yield of probe reduction only when the probe was intercalated into duplex DNA and charge was moving through the DNA. I then synthesized a methylated duplex that allowed for me to reversibly switch between the right-handed B-DNA and left-handed Z-DNA which allowed me to test the effect of supramolecular organization on chirality-induced spin selectivity while keeping the chirality of monomers constant. I found that the helicity of the DNA duplex determines which electron spin transports with higher yield through the DNA into an intercalated redox probe. This data shows that the three-dimensional organization of chiral monomers is more important than the chirality of those monomers for determining the spin selectivity of charge transport. This information has proved useful in the design of novel materials, and has been used to design cheaper organic material to replace inorganic magnets in devices.
“Helix-dependent Spin Filtering through the DNA Duplex” Zwang. T.J.; Hurlimann, S.; Hill, M.G.; Barton, J.K. J. Am. Chem. Soc., 2016, 138(48), 15551-15554
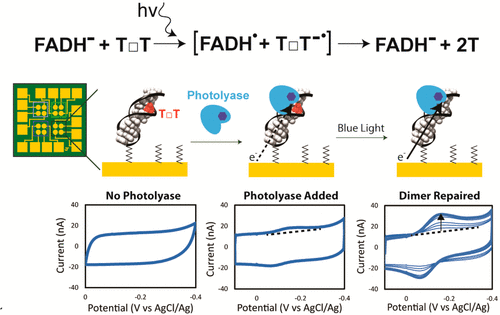
This exquisite spin selectivity begged the question: how might biology take advantage of such a spin filter? Photolyase and cryptochromes both have been shown to exhibit magnetosensitive chemistry nearby a DNA binding pocket, and photolyase had previously been shown capable of DNA CT. Thus, I conducted electrochemical studies to monitor the repair of cyclobutane pyrimidine dimer (CPD) lesions by E coli photolyase and truncated A Thaliana Cryptochrome 1 with an applied magnetic field. I found that the yield of dimer repair is dependent on the strength and angle of the applied magnetic field even when using magnetic fields weaker than 1 gauss. Further experiments show that the magnetic fields change the favorability of bond-breaking, and thus alter the efficiency of CPD repair. These data are the first radical pair found in biology that are sensitive to Earth’s magnetic field and, although there are still many questions to be answered, provides insight into a possible mechanism that animals may use to navigate with magnetic fields.
I now plan to use similar methodology to understand and modulate electrochemistry in vivo to provide insight into the complicated activities of neural systems.
"A Compass at weak Magnetic Fields using Thymine Dimer Repair” Zwang, T.J.; Tse, E.C.M.; Zhong, D.; Barton, J.K. ACS Cent. Sci., 2018, 4, 405-412